

The Precision Medicine Public-Private Partnership Alliance (PPP) coordinates with the clinical study platform (e.g., TCOG) to provide high-quality patient recruitment/data collection support to conduct the Collaborative Precision Medicine Research Program initiated and invested by the industrial partners. In this program, the LDTs certified NGS-based genetic tests are performed, with results applied to treatments and follow-up data collected. Also, patient consents to submit data to the National Biobank Consortium are obtained for future data use. Thus, the PPP project successfully links industrial, medical, academic, and government resources to achieve its four major goals: acceleration of clinical studies for new drug developments, enhancing cancer therapy developments to benefit patients, establishing cancer precision medicine databases conducive for R&D using RWD/RWE, and to facilitate evidence-based assessment for genetic tests insurance coverage policies.
醫療
Name:
Phone:
Address:No.488, Sec. 6, Zhongxiao E. Rd., Nangang Dist., Taipei City 115204, Taiwan (R.O.C.)

Degradable Implantable Flexible Microelectrode Patch with High-Resolution Electrical Stimulation, Electrophysiological Signal Sensing, and Cellular Regenerative Therapy
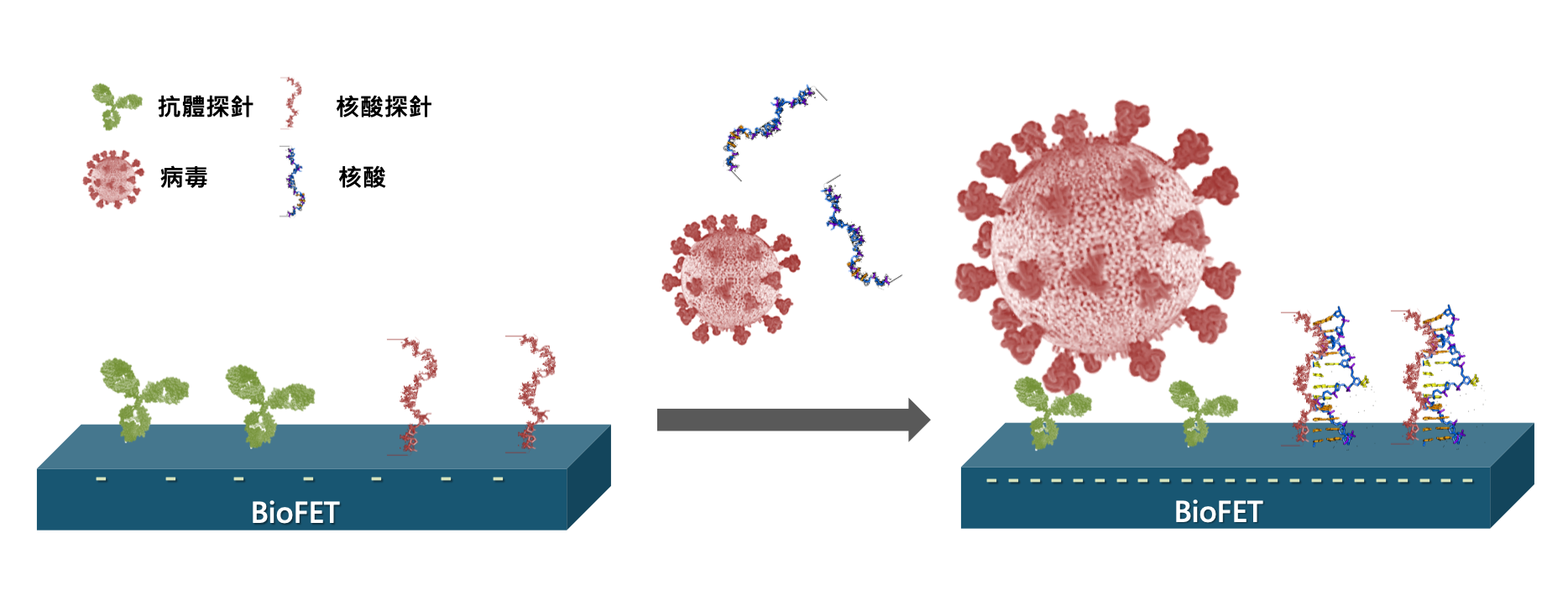
Ultrahigh-Sensitive Detection Platform Powered by Semiconductor-based Biosensor for the evaluation of Treatment on Acute Myeloid Leukemia
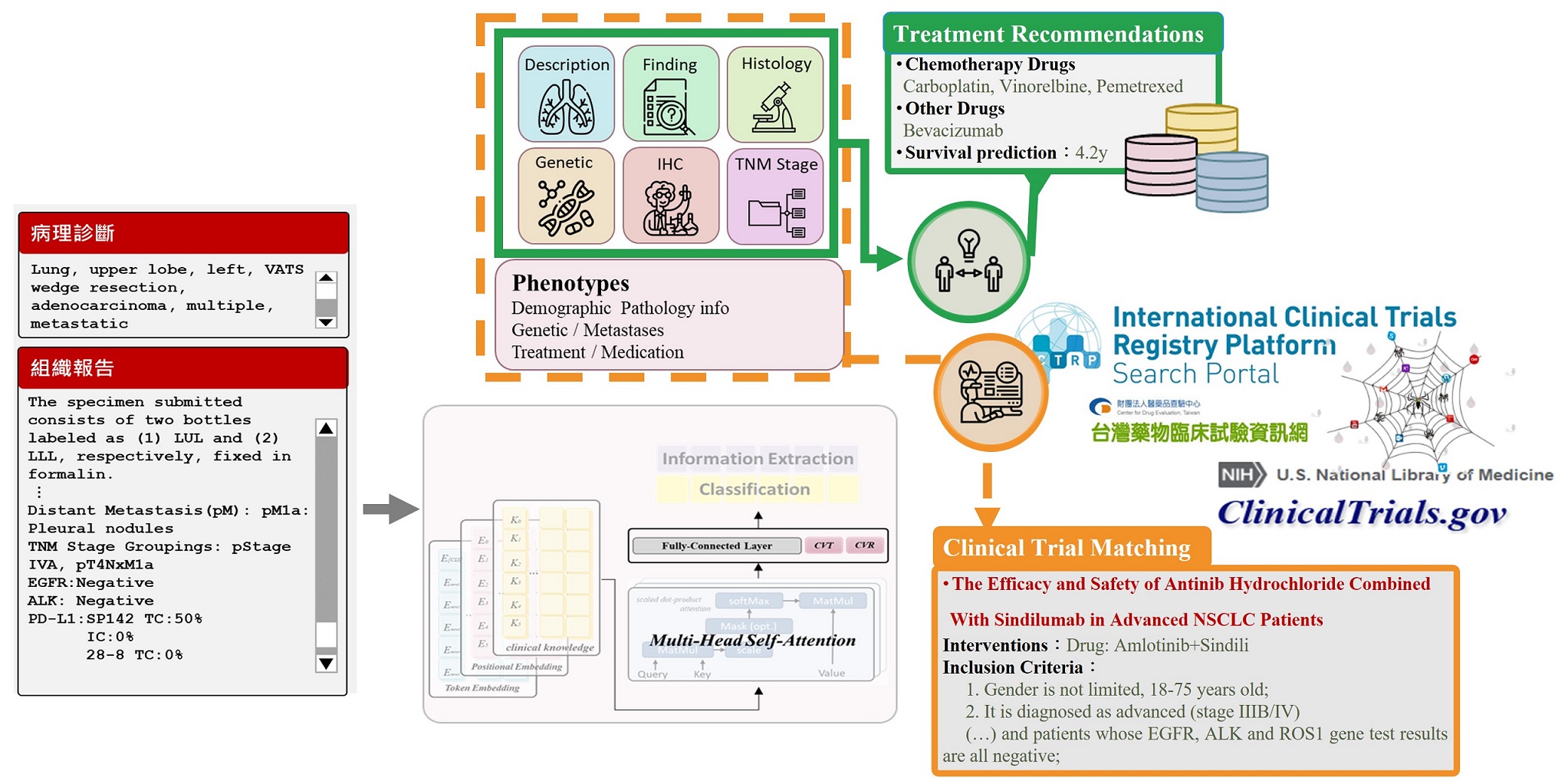
Using Generative Deep Learning to Predict Drug Response and Survival, a nd Automatically Match Clinical Trials for Advanced Lung Cancer.
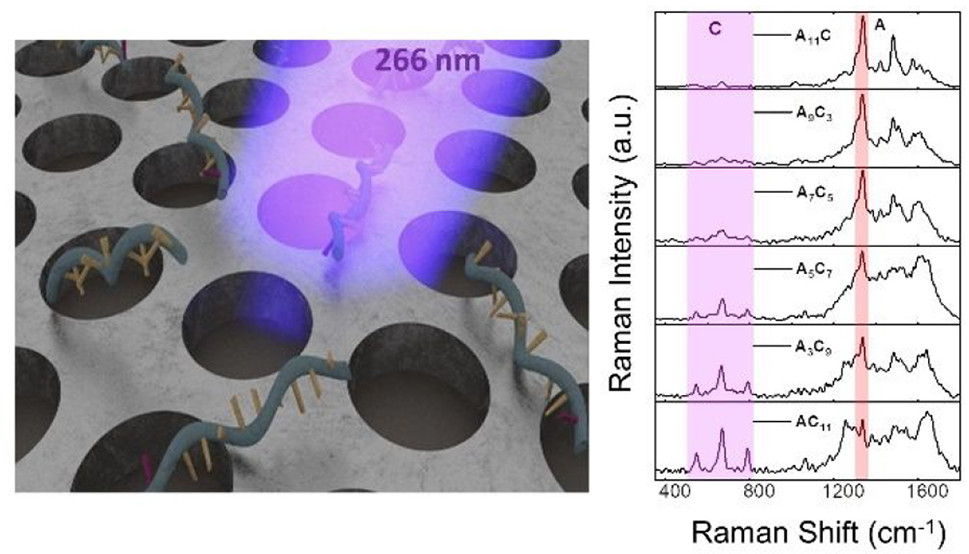
Demonstration of a Superior Deep-UV Surface-Enhanced Resonance Ram an Scattering (SERRS) Substrate and Single-Base Mutation Detection in O ligonucleotides
Technology maturity:Others
Exhibiting purpose:Product promotion
Trading preferences:Negotiate by self
Coming soon!